Your Cart is Empty
Have you ever walked down the eye care aisle at your local pharmacy or grocery store and noticed all of the artificial tears available? The choices seem infinite! So, how do you make the right choice?
First of all, you want to avoid preservatives in eye drops because preservatives can further irritate your already sensitive eyes. One of the major preservatives used in eye drops is benzalkonium chloride (BAK), and you'll find this in everything from artificial tears to allergy drops.
For many people, preservatives can be irritants so it's best to avoid them at all costs. Also, everyone has a different cause for their dryness, so everyone may react differently. Here are some other tips for choosing eye drops:
You're correct...this isn't an eye drop. However, we believe it's one of the most important products you can incorporate into your eye care routine, so we're making it #1. This hypochlorous acid cleanser is a great one for those of you with red, irritated eyes. We love this cleanser because it's helping to get to the root of the problem instead of just covering up the symptom, as most artificial tears do.
By cleansing with hypochlorous acid, you're supporting your eyelid health (and therefore your eye health).
This solution is super easy to use, there's no need to rinse it off, and it has a 2-year shelf life. Check price and reviews on Amazon!
We don't recommend eye drops often, but when we do it's usually these. We love this drop because they lubricate, moisten and support delicate eye tissues.
Oasis TEARS is a true solution that keeps tears on the eye surface. The drops are free of preservatives and come with 30 sterile, single-use vials per box. They're recommended for both day and nighttime use, and are safe for use with contact lenses.
We also have nothing but good things to say about this company and their mission. They are dedicated to patients, and you're only able to purchase these straight from your eye doctor. So, if you purchase them online (anywhere but our website), know that this goes against Oasis's doctor to patient model. Check price and reviews.
This new, preservative-free, low blur formula fortifies and restores the lipid layer of the tear film. Its unique blend of natural oils helps prevent tear evaporation and is clinically proven to support your eyes.
We love this drop because it provides the ocular surface with the healthy omega-3 oils we always speak about. MEGA-3 does not require shaking prior to use, comes in sterile vials, and is a great option we recommend. Check price and reviews on Amazon.
This artificial tear is a favorite of patients because it's been around awhile and it usually never disappoints. This is a non-preserved artificial tear that includes no additional chemicals which would irritate the eyes.
The goal of artificial tears is lubrication of the tissues on the ocular surface, and this is a great option for daily use. We used this artificial tear for years in our clinics, and it works well. Check price and reviews on Amazon.
This eye drop was a favorite of ours for many years, and we prescribed it often in our clinics. The preservative free Retaine MGD is a great choice at the end of a long day, but you can feel free to use these as often as you'd like since they contain no preservatives.
These also come in small vials for ease of carrying with you. You'll need to shake these well before use, so make note of that. Check price and reviews on Amazon.
Bausch and Lomb's Soothe Lubricant Eye Drops are clinically proven to soothe the eyes.These revolutionary, advanced drops provide soothing comfort and long lasting lubrication, keeping eyes feeling fresh throughout the day.
We recommend this eye drop because it helps maintain your eyes' natural moisture balance. And they're preservative free, so they're gentle enough to use as often as needed. Check price and reviews on Amazon.
This is a unique innovation in eye drops. NanoTears contains Nanopids, or tiny, clear, soothing lipids that revitalize the eyes' natural lubrication by restoring the lipid layer of the tear film. They provide a barrier to tear loss without the blur associated with other drops.
With these drops, you'll notice long-lasting hydration, and these come in single use vials similar to the drops mentioned above. Check price and reviews.
If you are still unsure on which drop to use, your local eye care provider can suggest specific drops just for you and your eye condition.
Now I'm going to discuss a bit about best practices for using artificial tears, no matter what type you choose from the above list. First, always wash your hands prior to putting eye drops in your eye (or your child’s). It does you no good to use eye drops to solve one problem if you inadvertently create an entirely new problem by introducing a new irritant into the eye. Also, make sure you don’t touch the dropper tip or eyeball with your hands, as you could introduce a foreign contaminant that would then be transferred to the eye.
Always use a mirror when inserting your eye drops; trying to freehand an eye drop into your eye creates a completely unnecessary risk that you will scratch your eye with the dropper tip or a fingernail.
Looking in the mirror, pull the lower eyelid down and away from the face, and place a drop (or more, if prescribed) into the gap you’ve created by pulling the eyelid down. Before moving on to the next eye, close the first eye and look around with the eye closed to evenly distribute the artificial tear around the inside of the eyelid. It may also help to press on the small puncta next to your nose, to keep drops from leaking out of your eyes.
Repeat the entire process on the other eye. Alternatively, feel free to lie back on a bed or couch and apply the drops if the above method proves to be too difficult.
Don’t panic if you feel an odd sensation, like tasting the eye drops or feeling them in the back of your throat; the various tubes and sinuses in your head are all interconnected, and it’s not uncommon to feel eye drops in your throat.
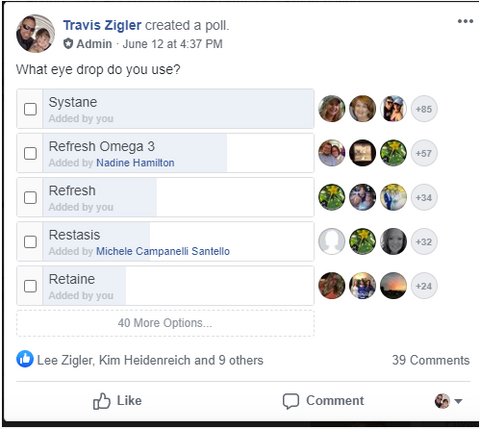
We decided to take a survey in our Support Community to find the 11 most used eye drops in hopes of simplifying your search. Below is a list of the results, including the product description, ingredients, directions, and where to purchase the drop.


Ingredients: Active Ingredients - Purpose.Polyethylene Glycol 400 0.4% - Lubricant.Propylene Glycol 0.3% - Lubricant. Inactive Ingredients: Boric Acid, Calcium Chloride, Hydroxypropyl Guar, Magnesium Chloride, Polyquaternium-1 as a preservative, Potassium Chloride, Purified Water, Sodium Chloride, Zinc Chloride.
Directions: Instill 1 or 2 drops in the affected eye(s) as needed. Other information: Store at room temperature.


Ingredients: Carboxymethylcellulose Sodium, Glycerine, Polysorbate 80
Directions: To open, twist and pull tab to remove. Instill 1 or 2 drops in the affected eye(s) as needed and discard the container.


Ingredients: Active Ingredients - Carboxymethylcellulose sodium 0.5%, Glycerin 1%, and Polysorbate 80 0.5%. Purpose: Eye lubricants. Inactive Ingredients - Boric acid; carbomer copolymer type A; castor oil; erythritol; levocarnitine; purified water; and sodium hydroxide.
Directions: To open, twist and pull tab to remove. Instill 1 or 2 drops in the affected eye(s) as needed and discard the container.
Restasis comes in both single dose vials and a new multidose bottle, and both act in the same way to produce the same results. It is recommended to make Restasis part of your daily routine and use it twice per day, morning and evening, for best results. From our experience in practice, it can often take patients six months or more to notice a change in their symptoms.
Allergan, the manufacturer of Restasis, offers a savings plan which allows you to save hundreds of dollars per month. In addition to this, they also offer assistance to those who qualify, and you could be eligible to get Restasis for free.


Ingredients: Light Mineral Oil (0.5%) Mineral Oil (0.5%)
Directions: Shake Well before using; Instill 1 or 2 drops in the affected eye(s) as needed and discard container.
Ingredients:0.2% Hyaluronic Acid
Directions:Recommended dosage is one drop in each eye. Apply the drops three times in a day. Drops can be used more frequently if required
Xiidra is a prescription eye drop approved by the FDA for both the signs and symptoms of dry, irritated eyes. Ask your eye doctor about prescription Xiidra.
Directions: To avoid contamination, make sure the container tip doesn’t touch anything. Tilt your head back looking skyward while gently pulling your lower eyelid down. Making sure the solution has moved to the tip of the Xiidra container, place the tip of the container close to (but not touching) your eye. Give the single-use container a gentle squeeze and drop into your eye. Repeat on the other side with the same container, and then discard.


Ingredients: Trehalose 3%, Sodium Hyaluronate 0.15%
Directions: Easy to squeeze bottle with no waste. Apply one drop in each eye.


Ingredients: Borate Buffers, Calcium Chloride, Dequest, Magnesium Chloride, Potassium Chloride, Purified Water, Sodium Bicarbonate, Sodium Chloride, Sodium Perborate, and Sodium Phosphate
Directions: Instill 1 or 2 drops in the affected eye(s) as needed. Do not use if imprinted neckband is broken or missing
In most cases, the more severe the dryness, the greater the need for autologous serum eye drops. Autologous serum eye drops are simply eye drops that have been made with the plasma that is located in your own blood. These drops are dependent solely on your blood and cannot be made with anything else.
This procedure and eye drop is not typically covered by medical insurance. To get a two month supply of autologous serum eye drops, you can expect to spend $175 to $300.
Vital Tears is an innovative company that is making it much easier for people to obtain autologous serum eye drops. This is great news for those who may be in rural areas or who may not have access to a specialist who is well versed in this technology.


Ingredients: Active Ingredients - Carboxymethylcellulose sodium 0.5%. Purpose: Eye lubricant. Inactive Ingredients - Calcium chloride; magnesium chloride; potassium chloride; purified water; sodium chloride; and sodium lactate. May also contain hydrochloric acid and/or sodium hydroxide to adjust pH.
Directions: To open, twist and pull tab to remove. Instill 1 or 2 drops in the affected eye(s) as needed and discard the container.
We, Eye Love, LLC, are a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for us to earn fees by linking to Amazon.com and affiliated sites. MediViz and Heyedrate branded products are made by our company, Eye Love, but we truly believe that our products are the best on the market! It is important to know that our products are not intended to diagnose, treat, cure, or prevent any disease.